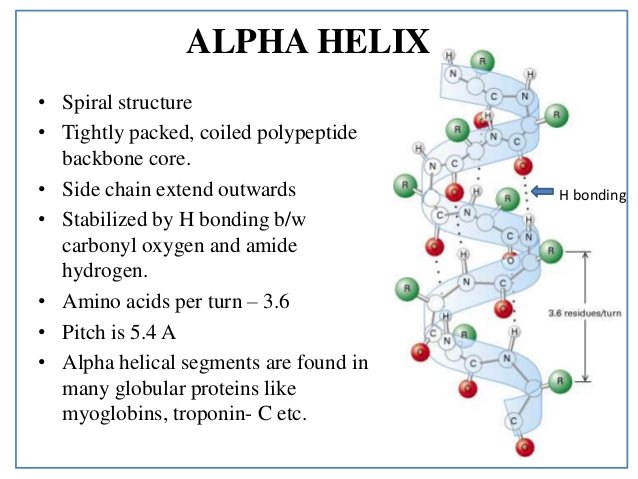
The C=O group of each peptide bond is bonded to the N-H group of the peptide bond forming a hydrogen bond. The C=O groups of all peptide bonds point in the opposite direction, and they are parallel to the axis of the helix. All the peptide bonds are trans and planar, and the N-H groups in the peptide bonds point in the same direction, which is approximately parallel to the axis of the helix. Features of the structure:Īlpha Helix: In alpha helix structure, there are 3.6 amino acids per turn of the helix. Examples of the structure:Īlpha Helix: Fingernails or toenails can be taken as an example of an alpha helix structure.īeta Pleated Sheet: The structure of feathers is similar to the structure of beta pleated sheet. It can happen in two ways parallel arrangement and anti-parallel arrangement. When two or more fragments of polypeptide chain(s) overlap with one another, forming a row of hydrogen bonds with each other, following structures can be found. Hydrogen bonds are indicated by the yellow dots. This spiral shape makes the alpha helix very strong. The hydrogen bonds allow the helix to hold the spiral shape and gives a tight coil. This enables all the amino acids in the chain to form hydrogen bonds (a bonding between an oxygen molecule and a hydrogen molecule) with each other. The formation of alpha helix structure happens when the polypeptide chains are twisted into a spiral. It is also known as the helicoidal arrangement of the peptide chain. In this structure, the polypeptide backbone is tightly bound around an imaginary axis as a spiral structure. What is the difference between Alpha Helix and Beta Pleated Sheet? Structure of Alpha Helix and Beta Pleated Sheet This structure allows forming more hydrogen bonds by stretching out the polypeptide chain. They are linked together by hydrogen bonds. It gets the name “pleated sheet” due to the wave-like appearance of the structure. In beta pleated sheets, the polypeptide chains run alongside each other. O atoms are red balls, N atoms are blue, and H atoms are omitted for simplicity sidechains are shown only out to the first sidechain C atom (green)ī) Edge-on view of the central two β strands Arrows indicate chain direction, and electron density contours outline the non-H atoms. The stability of alpha helix structure depends on several factors.Ĥ-stranded antiparallel β sheet fragment from a crystal structure of the enzyme catalase.Ī) showing the antiparallel hydrogen bonds (dotted) between peptide NH and CO groups on adjacent strands. This uniform pattern gives it definite features such as the thickness of the coil and it dictates the length of each complete turn along the helix axis. A hydrogen bond is formed per every four amino acid residues in the chain in the above manner. The hydrogen bonds formed between the oxygen of C=O group on the top coil and the hydrogen of the N-H group of the bottom coil help to hold the coil together. The range of amino acid residues can vary from 4 to 40 residues. What is Alpha Helix?Īn alpha helix is a right-handed coil of amino acid residues on a polypeptide chain. The key difference between Alpha Helix and Beta Pleated Sheet is in their structure they have two different shapes to do a specific job. These two structural components are the first main steps in the process of folding a polypeptide chain. Key Difference – Alpha Helix vs Beta Pleated SheetĪlpha helices and beta pleated sheets are the two most commonly found secondary structures in a polypeptide chain.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
